The PPAR-gamma agonist pioglitazone alleviates bleomycin-induced lung fibrosis in male BALB/c mice
Keywords:
pulmonary fibrosis, inflammation, bleomycin, pioglitazone, PPARGAbstract
Background: Many transcription factors may be involved in the pathogenesis of pulmonary fibrosis (PF). One such factor is PPARG. The PPARG agonist, pioglitazone (PG), has demonstrated general lung protective activity in animal models and is considered a promising therapeutic agent for fibrotic intervention. This study aimed to investigate the effect of PG on the expression of connective tissue remodeling genes in the lungs using bleomycin (BLM)-induced lung fibrosis (BIF).
Methods: The study was conducted on male BALB/c mice. PF was induced by pretreatment with bleomycin sulfate and cyclophosphamide. The mice were randomly divided into 3 groups: the first group was administered BLM at a dose of 0.15 U/kg in 50 µl of sterile saline (0.9% NaCl), and the second group was administered 50 µl of saline. The third group was not induced with pulmonary fibrosis and served as a control group. On the 4th day of the experiment, animals from the first and second groups were randomly divided into animals that received PG orally at a dose of 20 mg/kg or saline (40 µl) for 14 days. 4-hydroxyproline was determined in right lung tissue samples. Gene expression was determined using real-time PCR.
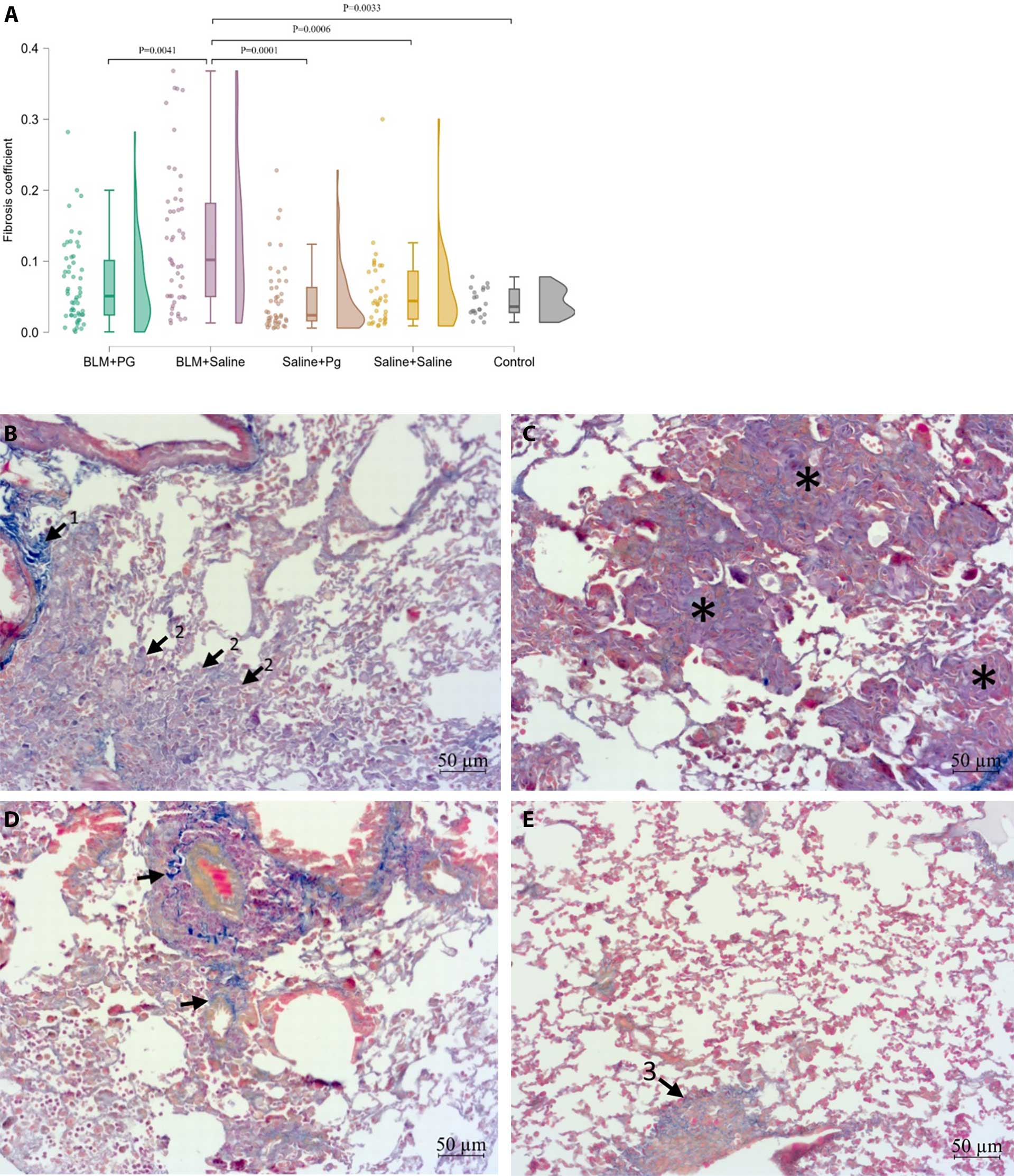
Results: PG administration to BIF mice resulted in a significant reduction and normalization of the total number of BAL cells, fibrosis score, mRNA expression of genes associated with connective tissue – Col1a1 (P=0.0466), Col3a1 (P=0.0053), Mmp2 (P=0.0006), Tgfb2 (P=0.0459), and Tgfb3 (P=0.0017), as well as mRNA of genes regulating lung connective tissue inflammation and fibrosis – Mrc1 (P=0.0263), Edn1 (P=0.0012), Pparg (P=0.0044), Nr1d1 (P=0.0053) and Fn1 (P=0.0125).
Conclusions: PPARG activation by PG alleviates BIF by decreasing collagen deposition, Col1a1 and Col3a1 mRNA expression, remodeling of lung tissue (decreasing Mmp2, Edn1, and Fn1 mRNA), and M2-specific profibrotic macrophage Mrc1 mRNA.
References
1. Stolz D, Mkorombindo T, Schumann DM, Agusti A, Ash SY, Bafadhel M, et al. Towards the elimination of chronic obstructive pulmonary disease: a Lancet Commission. Lancet 2022;400(10356):921-72.
2. Yang XY, Li F, Zhang G, Foster PS, Yang M. The role of macrophages in asthma-related fibrosis and remodelling. Pharmacol Ther 2025;269:108820.
3. Parimon T, Espindola M, Marchevsky A, Rampolla R, Chen P, Hogaboam CM. Potential mechanisms for lung fibrosis associated with COVID-19 infection. QJM 2023;116(7):487-92.
4. Muri J, Durcová B, Slivka R, Vrbenská A, Makovická M, Makovický P, et al. Idiopathic Pulmonary Fibrosis: Review of Current Knowledge. Physiol Res 2024;73(4):487-97.
5. Huo C, Jiao X, Wang Y, Jiang Q, Ning F, Wang J, et al. Silica aggravates pulmonary fibrosis through disrupting lung microbiota and amino acid metabolites. Sci Total Environ 2024;945:174028.
6. Xue S, Broerman MJ, Goobie GC, Kass DJ, Fabisiak JP, Wenzel SE, et al. Gaseous Air Pollutants and Lung Function in Fibrotic Interstitial Lung Disease (fILD): Evaluation of Different Spatial Analysis Approaches. Environ Sci Technol 2025;59(12):5936-45.
7. Weng C, Zhao Y, Song M, Shao Z, Pang Y, Yu C, et al. Mosaic loss of chromosome Y, tobacco smoking and risk of age-related lung diseases: insights from two prospective cohorts. Eur Respir J 2024;64(6):2400968.
8. Milman Krentsis I, Zheng Y, Rosen C, Shin SY, Blagdon C, Shoshan E, et al. Lung cell transplantation for pulmonary fibrosis. Sci Adv 2024;10(34):eadk2524.
9. George PM, Patterson CM, Reed AK, Thillai M. Lung transplantation for idiopathic pulmonary fibrosis. Lancet Respir Med 2019;7(3):271-82.
10. Fließer E, Jandl K, Lins T, Birnhuber A, Valzano F, Kolb D, et al. Lung Fibrosis Is Linked to Increased Endothelial Cell Activation and Dysfunctional Vascular Barrier Integrity. Am J Respir Cell Mol Biol 2024;71(3):318-31.
11. Khan MM, Galea G, Jung J, Zukowska J, Lauer D, Tuechler N, et al. Dextromethorphan inhibits collagen and collagen-like cargo secretion to ameliorate lung fibrosis. Sci Transl Med 2024;16(778):eadj3087.
12. Yadav P, Gómez Ortega J, Dabral P, Tamaki W, Chien C, Chang KC, et al. Myeloid-mesenchymal crosstalk drives ARG1-dependent profibrotic metabolism via ornithine in lung fibrosis. J Clin Invest 2025:e188734.
13. Wang J, Li K, Hao D, Li X, Zhu Y, Yu H, et al. Pulmonary fibrosis: pathogenesis and therapeutic strategies. MedComm (2020) 2024;5(10):e744.
14. Kamiya M, Carter H, Espindola MS, Doyle TJ, Lee JS, Merriam LT, et al. Immune mechanisms in fibrotic interstitial lung disease. Cell 2024;187(14):3506-30.
15. Alanazi FJ, Alruwaili AN, Aldhafeeri NA, Ballal S, Sharma R, Debnath S, et al. Pathological interplay of NF-κB and M1 macrophages in chronic inflammatory lung diseases. Pathol Res Pract 2025;269:155903.
16. Valenzi E, Jia M, Gerges P, Fan J, Tabib T, Behara R, et al. Altered AP-1, RUNX and EGR chromatin dynamics drive fibrotic lung disease. Preprint. bioRxiv 2024;2024.10.23.619858.
17. Wang Y, Zhao Y, Cao G, Jiang M, Yuan X, Li H, et al. STAT3 Facilitates Super Enhancer Formation to Promote Fibroblast-To-Myofibroblast Differentiation by the Analysis of ATAC-Seq, RNA-Seq and ChIP-Seq. J Cell Mol Med 2025;29(11):e70639.
18. Boateng E, Bonilla-Martinez R, Ahlemeyer B, Garikapati V, Alam MR, Trompak O, et al. It takes two peroxisome proliferator-activated receptors (PPAR-β/δ and PPAR-γ) to tango idiopathic pulmonary fibrosis. Respir Res 2024;25(1):345.
19. Pan X, Wang L, Yang J, Li Y, Xu M, Liang C, et al. TRβ activation confers AT2-to-AT1 cell differentiation and anti-fibrosis during lung repair via KLF2 and CEBPA. Nat Commun 2024;15(1):8672.
20. Wan R, Long S, Ma S, Yan P, Li Z, Xu K, et al. NR2F2 alleviates pulmonary fibrosis by inhibition of epithelial cell senescence. Respir Res 2024;25(1):154.
21. Wei A, Gao Q, Chen F, Zhu X, Chen X, Zhang L, et al. Inhibition of DNA methylation de-represses peroxisome proliferator-activated receptor-γ and attenuates pulmonary fibrosis. Br J Pharmacol 2022;179(7):1304-18.
22. Li G, Zhang Y, Jiang H, Wu X, Hao Y, Su Y, et al. PPARG/SPP1/CD44 signaling pathway in alveolar macrophages: Mechanisms of lipid dysregulation and therapeutic targets in idiopathic pulmonary fibrosis. Heliyon 2025;11(1):e41628.
23. Yang S, Sun Y, Luo Y, Liu Y, Jiang M, Li J, et al. Hypermethylation of PPARG-encoding gene promoter mediates fine particulate matter-induced pulmonary fibrosis by regulating the HMGB1/NLRP3 axis. Ecotoxicol Environ Saf 2024;272:116068.
24. Kökény G, Calvier L, Hansmann G. PPARγ and TGFβ-Major Regulators of Metabolism, Inflammation, and Fibrosis in the Lungs and Kidneys. Int J Mol Sci 2021;22(19):10431.
25. Amin F, Memarzia A, Roohbakhsh A, Shakeri F, Boskabady MH. Zataria multiflora and Pioglitazone Affect Systemic Inflammation and Oxidative Stress Induced by Inhaled Paraquat in Rats. Mediators Inflamm 2021;2021:5575059.
26. Legchenko E, Chouvarine P, Borchert P, Fernandez-Gonzalez A, Snay E, Meier M, et al. PPARγ agonist pioglitazone reverses pulmonary hypertension and prevents right heart failure via fatty acid oxidation. Sci Transl Med 2018;10(438):eaao0303.
27. Kurihara C, Sakurai R, Chuang TD, Waring AJ, Walther FJ, Rehan VK. Combination of pioglitazone, a PPARγ agonist, and synthetic surfactant B-YL prevents hyperoxia-induced lung injury in adult mice lung explants. Pulm Pharmacol Ther 2023;80:102209.
28. Tang J, Dong W, Wang D, Deng Q, Guo H, Xiao G. Upregulation of PGC-1α expression by pioglitazone mediates prevention of sepsis-induced acute lung injury. Braz J Med Biol Res 2024;57:e13235.
29. Yeligar SM, Mehta AJ, Harris FL, Brown LAS, Hart CM. Pioglitazone Reverses Alcohol-Induced Alveolar Macrophage Phagocytic Dysfunction. J Immunol 2021;207(2):483-92.
30. Huang T, Lin R, Su Y, Sun H, Zheng X, Zhang J, et al. Efficient intervention for pulmonary fibrosis via mitochondrial transfer promoted by mitochondrial biogenesis. Nat Commun 2023;14(1):5781.
31. Schrier DJ, Phan SH. Modulation of bleomycin-induced pulmonary fibrosis in the BALB/c mouse by cyclophosphamide-sensitive T cells. Am J Pathol 1984;116(2):270-8.
32. Egger C, Cannet C, Gérard C, Jarman E, Jarai G, Feige A, et al. Administration of bleomycin via the oropharyngeal aspiration route leads to sustained lung fibrosis in mice and rats as quantified by UTE-MRI and histology. PLoS One 2013;8(5):e63432.
33. Kabaley A, Izmailova O, Palchyk V, Shinkevich V, Shlykova O. The PPARG receptor agonist pioglitazone reduces the manifestations of pulmonary fibrosis in mice induced by bleomycin administration. Med and Ecol probl 2024;28(3):19-27.
34. Shlykova O, Izmailova O, Kabaliei A, Palchyk V, Shynkevych V, Kaidashev I. PPARG stimulation restored lung mRNA expression of core clock, inflammation- and metabolism-related genes disrupted by reversed feeding in male mice. Physiol Rep 2023;11(17):e15823.
35. Van Hoecke L, Job ER, Saelens X, Roose K. Bronchoalveolar lavage of murine lungs to analyze inflammatory cell infiltration. J Vis Exp 2017;(123):55398.
36. Piccinini F, Tesei A, Arienti C, Bevilacqua A. Cell counting and viability assessment of 2D and 3D cell cultures: expected reliability of the Trypan Blue assay. Biol Proced Online 2017;19:8.
37. Memon MA, Anway MD, Covert TR, Uzumcu M, Skinner MK. Transforming growth factor beta (TGFbeta1, TGFbeta2 and TGFbeta3) null-mutant phenotypes in embryonic gonadal development. Mol Cell Endocrinol 2008;294(1-2):70-80.
38. Lu J, Xie L, Liu C, Zhang Q, Sun S. PTEN/PI3k/AKT Regulates Macrophage Polarization in Emphysematous mice. Scand J Immunol 2017;85(6):395-405.
39. Neder TH, Schrankl J, Fuchs MAA, Broeker KAE, Wagner C. Endothelin receptors in renal interstitial cells do not contribute to the development of fibrosis during experimental kidney disease. Pflugers Arch 2021;473(10):1667-83.
40. Ke L, Yang Y, Li JW, Wang B, Wang Y, Yang W, et al. Modulation of Corneal FAK/PI3K/Akt Signaling Expression and of Metalloproteinase-2 and Metalloproteinase-9 during the Development of Herpes Simplex Keratitis. Biomed Res Int 2019;2019:4143981.
41. Kabaliei A, Palchyk V, Izmailova O, Shynkevych V, Shlykova O, Kaidashev I. Long-term administration of Omeprazole-induced hypergastrinemia and changed glucose homeostasis and expression of metabolism-related genes. Biomed Res Int 2024;2024:7747599.
42. Liu S, Cai Y, Sothern RB, Guan Y, Chan P. Chronobiological analysis of circadian patterns in transcription of seven key clock genes in six peripheral tissues in mice. Chronobiol Int 2007;24(5):793-820.
43. Masugi Y, Abe T, Tsujikawa H, Effendi K, Hashiguchi A, Abe M, et al. Quantitative assessment of liver fibrosis reveals a nonlinear association with fibrosis stage in nonalcoholic fatty liver disease. Hepatol Commun 2017;2(1):58-68.
44. Moore BB, Hogaboam CM. Murine models of pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 2008;294(2):L152-L160.
45. Adamcakova J, Palova R, Mokra D. Experimental models of pulmonary fibrosis and their translational potential. Acta Medica Martiniana 2019;19(3):95-102.
46. Li X, Sun X, Kan C, Chen B, Qu N, Hou N, et al. COL1A1: A novel oncogenic gene and therapeutic target in malignancies. Pathol Res Pract 2022;236:154013.
47. Lin Y, Li Y, Chen P, Zhang Y, Sun J, Sun X, et al. Exosome-based regimen rescues endometrial fibrosis in intrauterine adhesions via targeting clinical fibrosis biomarkers. Stem Cells Transl Med 2023;12(3):154-68.
48. Chen P, Xie L, Ma L, Zhao X, Chen Y, Ge Z. Prediction and analysis of genetic effect in idiopathic pulmonary fibrosis and gastroesophageal reflux disease. IET Syst Biol 2023;17(6):352-65.
49. Wang R, Wu G, Dai T, Lang Y, Chi Z, Yang S, et al. Naringin attenuates renal interstitial fibrosis by regulating the TGF-β/Smad signaling pathway and inflammation. Exp Ther Med 2021;21(1):66.
50. Zhao J, Yu W, Zhou D, Liu Y, Wei J, Bi L, et al. Delineating, imaging, and assessing pulmonary fibrosis remodeling via collagen hybridization. ACS Nano 2024;18(41):27997-28011.
51. Song S, Fu Z, Guan R, Zhao J, Yang P, Li Y, et al. Intracellular hydroxyproline imprinting following resolution of bleomycin-induced pulmonary fibrosis. Eur Respir J 2022;59(5):2100864.
52. Sun T, Huang Z, Liang WC, Yin J, Lin WY, Wu J, et al. TGFβ2 and TGFβ3 isoforms drive fibrotic disease pathogenesis. Sci Transl Med 2021;13(605):eabe0407.
53. Duan FF, Barron G, Meliton A, Mutlu GM, Dulin NO, Schuger L. P311 promotes lung fibrosis via stimulation of transforming growth factor-β1, -β2, and -β3 translation. Am J Respir Cell Mol Biol 2019;60(2):221-31.
54. Pommerolle L, Beltramo G, Biziorek L, Truchi M, Dias AMM, Dondaine L, et al. CD206+ macrophages are relevant non-invasive imaging biomarkers and therapeutic targets in experimental lung fibrosis. Thorax 2024;79(12):1124-35.
55. Swigris JJ, Brown KK. The role of endothelin-1 in the pathogenesis of idiopathic pulmonary fibrosis. BioDrugs 2010;24(1):49-54.
56. Alsafadi HN, Staab-Weijnitz CA, Lehmann M, Lindner M, Peschel B, Königshoff M, et al. An ex vivo model to induce early fibrosis-like changes in human precision-cut lung slices. Am J Physiol Lung Cell Mol Physiol 2017;312(6):L896-L902. Erratum in: Am J Physiol Lung Cell Mol Physiol 2020;318(4):L844.
57. Guo Z, Wu J, Zhao J, Liu F, Chen Y, Bi L, et al. [IL-33/ST2 promotes airway remodeling in asthma by activating the expression of fibronectin 1 and type 1 collagen in human lung fibroblasts]. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2014;30(9):975-9.
58. Zhang-Sun ZY, Xu XZ, Escames G, Lei WR, Zhao L, Zhou YZ, et al. Targeting NR1D1 in organ injury: challenges and prospects. Mil Med Res 2023;10(1):62.
59. Malur A, Mohan A, Barrington RA, Leffler N, Malur A, Muller-Borer B, et al. Peroxisome proliferator-activated receptor-γ deficiency exacerbates fibrotic response to mycobacteria peptide in murine sarcoidosis model. Am J Respir Cell Mol Biol 2019;61(2):198-208.
60. Zhao Z, Shan X, Ding J, Ma B, Li B, Huang W, et al. Boosting RNA nanotherapeutics with V-ATPase activating non-inflammatory lipid nanoparticles to treat chronic lung injury. Nat Commun 2025;16(1):6477.
61. Raza GS, Sodum N, Kaya Y, Herzig KH. Role of circadian transcription factor Rev-Erb in metabolism and tissue fibrosis. Int J Mol Sci 2022;23(21):12954.
62. Esmaeili S, Salari S, Kaveh V, Ghaffari SH, Bashash D. Alteration of PPAR-GAMMA (PPARG; PPARγ) and PTEN gene expression in acute myeloid leukemia patients and the promising anticancer effects of PPARγ stimulation using pioglitazone on AML cells. Mol Genet Genomic Med 2021;9(11):e1818.
63. Costa V, Gallo MA, Letizia F, Aprile M, Casamassimi A, Ciccodicola A. PPARG: gene expression regulation and next-generation sequencing for unsolved issues. PPAR Res 2010;2010:409168.
64. Nielsen R, Pedersen TA, Hagenbeek D, Moulos P, Siersbaek R, Megens E, et al. Genome-wide profiling of PPARgamma: RXR and RNA polymerase II occupancy reveals temporal activation of distinct metabolic pathways and changes in RXR dimer composition during adipogenesis. Genes Dev 2008;22(21):2953-67.
65. Lefterova MI, Steger DJ, Zhuo D, Qatanani M, Mullican SE, Tuteja G, et al. Cell-specific determinants of peroxisome proliferator-activated receptor gamma function in adipocytes and macrophages. Mol Cell Biol 2010;30(9):2078-89.
66. Fontaine C, Dubois G, Duguay Y, Helledie T, Vu-Dac N, Gervois P, et al. The orphan nuclear receptor Rev-Erbalpha is a peroxisome proliferator-activated receptor (PPAR) gamma target gene and promotes PPARgamma-induced adipocyte differentiation. J Biol Chem 2003;278(39):37672-80.
67. Levine B. Eating oneself and uninvited guests: autophagy-related pathways in cellular defense. Cell 2005;120(2):159-162.
68. Manzéger A, Garmaa G, Mózes MM, Hansmann G, Kökény G. Pioglitazone Protects Tubular Epithelial Cells during Kidney Fibrosis by Attenuating miRNA Dysregulation and Autophagy Dysfunction Induced by TGF-β. Int J Mol Sci 2023;24(21):15520.
69. Gao D, Ning N, Hao G, Niu X. Pioglitazone attenuates vascular fibrosis in spontaneously hypertensive rats. PPAR Res 2012;2012:856426.
70. Zhang J, Huang X, Wang L. Pioglitazone inhibits the expression of matrix metalloproteinase-9, a protein involved in diabetes-associated wound healing. Mol Med Rep 2014;10(2):1084-1088.
71. Aluru S, Thyagarajan A, Sahu RP. Pioglitazone-Based Combination Approaches for Non-Small-Cell Lung Cancer. Pharmaceutics 2025;17(11):1416.
72. Serizawa M, Murakami H, Watanabe M, Takahashi T, Yamamoto N, Koh Y. Peroxisome proliferator-activated receptor γ agonist efatutazone impairs transforming growth factor β2-induced motility of epidermal growth factor receptor tyrosine kinase inhibitor-resistant lung cancer cells. Cancer Sci 2014;105(6):683-9.
73. Redondo S, Hristov M, Gümbel D, Tejerina T, Weber C. Biphasic effect of pioglitazone on isolated human endothelial progenitor cells: involvement of peroxisome proliferator-activated receptor-gamma and transforming growth factor-beta1. Thromb Haemost 2007;97(6):979-87.
74. Yu Q, Cheng P, Wu J, Guo C. PPARγ/NF-κB and TGF-β1/Smad pathway are involved in the anti-fibrotic effects of levo-tetrahydropalmatine on liver fibrosis. J Cell Mol Med 2021;25(3):1645-60.
75. Koo JB, Nam MO, Jung Y, Yoo J, Kim DH, Kim G, et al. Anti-fibrogenic effect of PPAR-γ agonists in human intestinal myofibroblasts. BMC Gastroenterol 2017;17(1):73.
76. Stow LR, Jacobs ME, Wingo CS, Cain BD. Endothelin-1 gene regulation. FASEB J 2011;25(1):16-28.
77. Yun Z, Maecker HL, Johnson RS, Giaccia AJ. Inhibition of PPAR gamma 2 gene expression by the HIF-1-regulated gene DEC1/Stra13: a mechanism for regulation of adipogenesis by hypoxia. Dev Cell 2002;2(3):331-41.
78. Xu Y, Takahashi Y, Wang Y, Hama A, Nishio N, Muramatsu H, et al. Downregulation of GATA-2 and overexpression of adipogenic gene-PPARgamma in mesenchymal stem cells from patients with aplastic anemia. Exp Hematol 2009;37(12):1393-9.
79. Carvalho MV, Gonçalves-de-Albuquerque CF, Silva AR. PPAR Gamma: From Definition to Molecular Targets and Therapy of Lung Diseases. Int J Mol Sci 2021;22(2):805.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 The Author(s)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Mattioli 1885 has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.




